While most of us are familiar with the warm glow of traditional incandescent bulbs, many may not be aware of how this type of bulb actually works. Here’s a bit of background: incandescent bulbs produce light by heating a filament till it glows – in fact, up to 90% of the energy used to light an incandescent bulb goes towards producing this heat!
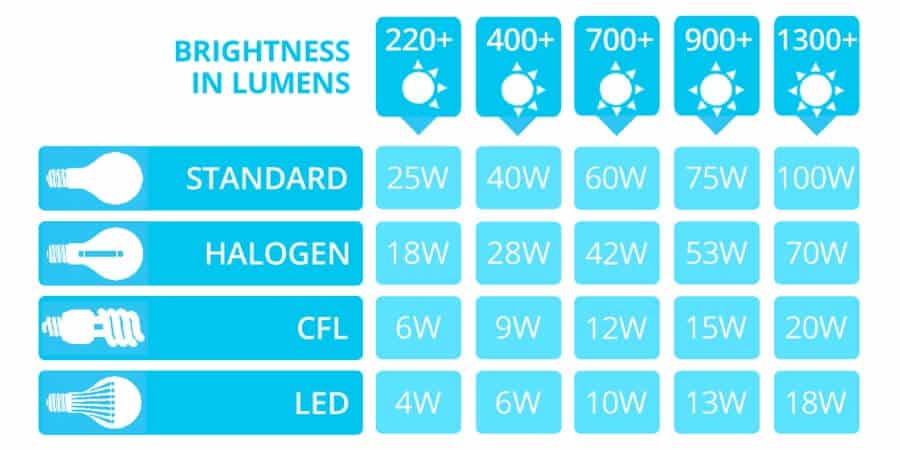
Their high energy usage is just one reason why many governments have begun phasing out traditional light bulbs in favour of energy efficient alternatives like CFLs and LEDs. But for those who are unfamiliar with modern lighting options like LEDs, making the switch to a new style of light bulb can seem like a daunting prospect. Understanding what LEDs are and how they work can ease the process of finding the right bulb and switching to energy efficient lighting. Keep reading to learn more.
What is an LED?
“LED” stands for “light emitting diode”. The first two parts of this name are easy to understand – “light emitting” means that it gives off light – but what exactly is a “diode”, and what does it do? To answer that question, we have to get a bit scientific.
All diodes emit photons (particles of electromagnetic energy), but only certain types of diodes emit that electromagnetic energy as light instead of heat. A light emitting diode is a type of solid-state lighting (SSL) technology, meaning that it emits light from a piece of solid matter. In this case, that piece of solid matter is a two-lead . We’ll explain how this works in the next section.
Types of LEDs
There are two basic types of LEDs:

- Indicator – an inexpensive, low-power device that is used as indicator lights in cars, panels, and other electronic devices.

- Illuminator – a high-power device that provides illumination. These are the type of LEDs you buy to illuminate rooms in your home or office. They can be found in a variety of styles, shapes and colours suitable for almost any application.
How do LEDs work?
An LED is capable of generating light because of the arrangement of the two semiconductor materials located between its electrodes:
- N-type: A semiconductor with extra electrons (also known as extra negatively charged particles).
- P-type: A semiconductor with extra holes (also known as extra positively charged particles).
Connecting the N-type semiconductor to the negative electrode and the P-type semiconductor to the positive electrode activates the electrons so they can flow across the junction from the negative to the positive layer. As the extra electrons (negatively charged particles) move through the extra holes (positively charged particles), they emit light.
![]()
The conductive material that makes up LEDs is typically aluminum-gallium-arsenide (AlGaAs), but there are other kinds. The materials are selected specifically because they produce photons that will be released on the visible portion of the light spectrum. The type of material chosen and the amount used alters the colour of the light because each material generates photons at different wavelengths, affecting how it appears to the human eye.
Read More
Find answers to all your questions about LEDs in the rest of our Ultimate Guide to LED Lights:
Part 1: What is an LED and How Do LEDs Work? (you’re here!)
Part 2: Advantages of LEDs
Part 3: LEDs vs. Traditional Incandescent Lights
Part 4: Switching to LEDs in 5 Steps
Part 5: How to Buy LEDs
Part 6: History of LEDs
Part 7: Advanced Features